by Eduardo Libby
Versión en Español en inorganicucr.com
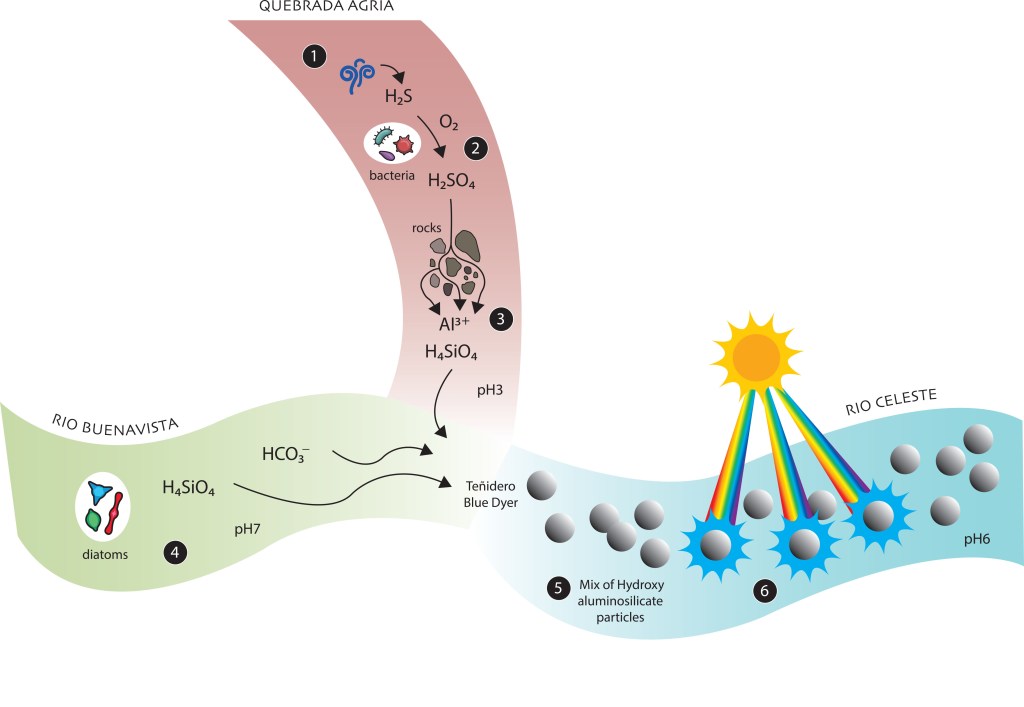
The Rio Celeste is formed at the junction of two mountain rivers with crystalline waters that originate on the slopes of Tenorio Volcano, in Northern Costa Rica. One is clean water Buenavista River. The other is Quebrada Agria (Sour Creek) which owes its name to the fact that it contains dissolved sulfuric acid, aluminum and silica. When the two streams come together at the site known locally as the “Teñidero,” (Dyeing Shop) the mixture produces a suspension of particles with sizes less than a thousandth of a millimeter that scatters the blue component of sunlight and gives Río Celeste its beautiful turquoise color.
Although very clean water sometimes appears blue in the ocean, rivers and lakes, this is because water can slightly absorb light in the red part of the spectrum and lets the blue part through. However, this effect is very weak and is only noticeable when the water is several meters deep. The most common, however, is that due to the presence of algae or suspended mineral particles, the water of many rivers looks greenish, reddish or brown.
When water looks turquoise in waters of glacial origin or in volcanic regions such as Rio Celeste, we also notice that it is not completely transparent due to suspended material and its color is due to Mie Scattering. It receives this name in honor of the German physicist Gustav Mie who was the first to describe the mathematics of the phenomenon. The exact color of the scattered light depends on the size of the particles and the light source, but it is usually a blue-green or cyan color, close to what we know as “celeste” in Spanish. The blue (actually cyan) color of the sky is also due to the scattering of light, in this case by molecules in the atmosphere and is called Rayleigh scattering which is a special case of Mie scattering when the particles are much smaller than the wavelength of light.

In glacial waters and in the Rio Celeste, visible light falls on particles with a size comparable to its wavelength, which is 0.4 to 0.7 micrometers (thousandths of a millimeter). In these cases, the reddish component of the spectrum, of longer wavelengths, continues on its way but the bluish component deviates in all directions, that is, it is scattered. As the rays are scattered in all directions, part of the light we see directly from the above the water surface but another part is in turn reflected by other particles towards us. In the meltwater of glaciers this is produced by finely ground rock particles called “Glacial Flour.” In Costa Rica we do not have glaciers but the lake of Poas Volcano’s active crater shows a similar light-blue color due to Mie scattering of light in this case by colloidal sulfur particles. Let us note that in the scattering of light the color does not correspond to that of the material of the particles because they are not large enough to be directly observed. For example, during our investigations we have found that Río Celeste particles are in fact a complex mixture of colorless silicon and aluminum minerals.

To further understand the color phenomenon of Rio Celeste we would have to delve into Mie’s Theory of electromagnetic wave scattering. We will define scattering as the change in direction that waves experience when they encounter in their path obstacles of a size comparable to their wavelength. An everyday example of scattering are sound waves that change their direction at doors and that is why we can hear sounds from other rooms even if we do not have a direct line of sight to the source. The mathematics of the Theory are complicated but the theory successfully explains all scattering processes from the color of the sky to the operation of Radar. In the references there is an article illustrating the change in color of scattered light as a function of particle size and the more physically and mathematically inclined can then read it and learn more about it on the internet.

Image. In the “Blue Pond” you can see that water of Rio Celeste is a little greener than the color of the sky. The exact observed color depends on environmental factors such as the color of the river bottom and the power spectrum of the illuminant, in this case the direct Sun.
Initially we managed to explain the color of Rio Celeste based on a mathematical model of Mie scattering developed by Dr. William Vargas from the School of Physics and CICIMA, University of Costa Rica, based on careful measurements of the size distribution and properties of the particles that form in the Teñidero made by Drs. Erick Castellón (School of Chemistry and CICIMA, UCR and Dr. Max Chavarría (also from the School of Chemistry, CIPRONA, UCR, and CENIBIOT) together with researchers from Costa Rica National University.

Our subsequent work has extended these initial results, giving us a clearer idea of how the particles are formed in the Teñidero. We now know that what occurs is a chemical reaction between the soluble species of sulfuric acid, silicic acid and aluminum from Quebrada Agria with the alkaline ions from Buenavista River. The reaction rapidly generates the colloidal aluminosilicate particles that scatter blue light in Río Celeste.
But, where do the acidity and ions of Quebrada Agria come from? The answer comes from other work we have done on the Río Roble, the tributary that joins the Rio Celeste at Laguna Azul (Blue Pond). Dr. Max Chavarría and his collaborators found that in the hydrothermal water and gas vents we see at Los Borbollones, underground hydrogen sulfide is oxidized to sulfuric acid by microorganisms:
H2S + 2 O2 → H2SO4 + energy
These are sulfur-oxidizing bacteria of the genera Sulfuriferula, Halothiobacillus and Sulfurimonas that take advantage of the chemical oxidation of sulfur with oxygen to obtain metabolic energy. The sulfuric acid produced by this reaction attacks the rocks, mainly dissolving the feldspars, with which the aluminum and silicon pass into the aqueous phase as Al3+ ions and silicic acid (H4SiO4).


The Quebrada Agria (Sour Creek) originates as a common mountain river but comes into contact with hydrothermal vents such as those of Roble River which turn it into an acid current with a pH of 3-4 (an acidity between that of lemon juice and that of orange juice) due to the reaction with the rocks of the riverbed. The Buenavista River, with a pH of 7-7.5, like clean mountain rivers, contains basic bicarbonate ions (HCO3–) that can neutralize the acidity of sulfuric acid. In addition, the Buenavista River contains silicic acid, a product of the dissolution of siliceous shells of its abundant diatoms and also of the soils rich in volcanic glass on the slopes of Tenorio Volcano. The two currents resulting mixture acquires an intermediate pH of 5-6. At these pH values solid aluminum hydroxides begin to form but in the presence of silicic acid they quickly become a colloid of hydroxyaluminosilicate particles that scatter blue light.
The chemistry of Rio Celeste :
First, bicarbonate ions from the Buenavista River neutralize sulfuric acid from the Quebrada Agria:
2 HCO3– + H2SO4 → SO42- + 2 H2O + 2 CO2
But the Buenavista River is fast enough to leave behind unreacted bicarbonate, which produces hydroxide ions.
HCO3– + H2O → H2CO3 + OH–
Hydroxide ions react with aluminum ions from Quebrada Agria forming
Al(OH)3
2 Al3+ + 6 OH– → 2 Al(OH)3
Finally, the silicic acid reacts with the aluminum hydroxide generating hydroxyaluminosilicates of approximate composition
(Al2O3)(SiO2)2•3H2O as amorphous particles due to their rapid formation rate.
2 H4SiO4 + 2 Al(OH)3 → (Al2O3)(SiO2)2•3H2O + 4 H2O

Note that if there were no silicic acid in Quebrada Agria then hydroxyaluminosilicates would not form. This is an important difference with respect to acidic rivers in other regions.
Hydroxyaluminosilicates belong to a group of minerals that do not form crystals and are known as Short-Range Ordered Aluminosilicates (SROAS). Their chemical bonds are similar to those of Imogolite and Allophane SROAS and they are found in volcanic soils called Andosols that are common in Costa Rica. Imogolite and Allophane have definite shapes of tubes and hollow spheres respectively. The hydroxyaluminosilcates for their part do not have such a defined structure.
Our group from the University of Costa Rica took water samples from the Río Celeste and isolated the particles by filtration through a very fine membrane. Next, we characterized the particles using Electron Microscopy, Powder X-ray Diffraction and Infrared Spectroscopy techniques at the School of Chemistry and at the Center for Materials Science and Engineering Research (CICIMA). By means of a comparison with the properties of synthetic SROAS just recently reported in the literature, we were able to verify that indeed the particles of the Río Celeste are recently formed hydroxyaluminosilicates. In the references you can consult the work that we did together with international collaborators in Germany and Sweden for the study of the microbial communities involved.
Hydroxyaluminosilicates are of great environmental importance as they keep aluminum ions, toxic to life, trapped within a highly insoluble solid, separating them from drinking water. The environmental chemistry of silicon and aluminum are thus closely linked and affect all biological systems.

We owe our success in the study and exploration of the Río Celeste system to the support of the National System of Conservation Areas (SINAC) and especially to the energy and enthusiasm of Master Isaac López Núñez who currently manages Tenorio Volcano National Park. We are deeply grateful to him and the Park staff for their support.
We conclude by mentioning that in Costa Rica we also find rivers with blue waters on the slopes of Poás and Rincón de La Vieja volcanoes and we are also investigating them. These other blue rivers in Costa Rica are just as beautiful but they take on their color slowly, over several kilometers. As far as we know, they do not seem to have a Teñidero like the one on Río Celeste. Observing two colorless streams that join in the middle of the tropical forest and instantly turn into a river with waters dyed the color of Heaven is a natural miracle that Costa Ricans must witness and admire.

Further Reading
Arce-Rodríguez, A.; Libby, E.; Castellón, E.; Avendaño, R.; Cambronero, J. C.; Vargas, M.; Pieper, D. H.; Bertilsson, S.; Chavarría, M.; Puente-Sánchez, F. Out of the Blue: The Independent Activity of Sulfur-Oxidizers and Diatoms Mediate the Sudden Color Shift of a Tropical River. Environmental Microbiome 2023, 18 (1), 6. https://doi.org/10.1186/s40793-023-00464-2. – Our recent work on the characterization of Río Celeste particles and their formation mechanism. The main source for this post.
Beardmore, J.; Lopez, X.; Mujika, J. I.; Exley, C. What Is the Mechanism of Formation of Hydroxyaluminosilicates? Scientific Reports 2016, 6 (1), 30913. https://doi.org/10.1038/srep30913. – A description of the initial stages of formation of hydroxyaluminosilicates.
Exley, C.; Guerriero, G.; Lopez, X. Silicic Acid: The Omniscient Molecule. Science of The Total Environment 2019, 665, 432–437. https://doi.org/10.1016/j.scitotenv.2019.02.197. – Silicon and aluminum in biological systems. References to the authors previous work on this topic.
Levard, C.; Doelsch, E.; Basile-Doelsch, I.; Abidin, Z.; Miche, H.; Masion, A.; Rose, J.; Borschneck, D.; Bottero, J.-Y. Structure and Distribution of Allophanes, Imogolite and Proto-Imogolite in Volcanic Soils. Geoderma 2012, 183–184, 100–108. https://doi.org/10.1016/j.geoderma.2012.03.015. – SROAS formation and discussion of tetrahedral and octahedral aluminum sites in their structure.
Arce-Rodríguez, A.; Puente-Sánchez, F.; Avendaño, R.; Martínez-Cruz, M.; de Moor, J. M.; Pieper, D. H.; Chavarría, M. Thermoplasmatales and Sulfur-Oxidizing Bacteria Dominate the Microbial Community at the Surface Water of a CO2-Rich Hydrothermal Spring Located in Tenorio Volcano National Park, Costa Rica. Extremophiles 2019, 23 (2), 177–187. https://doi.org/10.1007/s00792-018-01072-6. – Source of water’s acidity in Los Borbollones.
Gedzelman, S. D. Simulating Colors of Clear and Partly Cloudy Skies. Applied Optics 2005, 44 (27), 5723. https://doi.org/10.1364/AO.44.005723. – This is an article about light scattering by different particle sizes. Although it refers to the atmosphere, the basic principles are the same. Figure 3 is particularly explanatory. There is also a simplified explanation on this website:
https://geo.libretexts.org/Bookshelves/Meteorology_and_Climate_Science/Practical_Meteorology_(Stull)/22%3A_Atmospheric_Optics/22.03%3A_New_Page
Causes of Color is an excellent website that has a page dedicated to explaining why water is intrinsically blue. https://www.webexhibits.org/causesofcolor/5B.html
